 Have a look at Maxwell's Demon for one of the more slippery problems with entropy, which links it squarely to information. 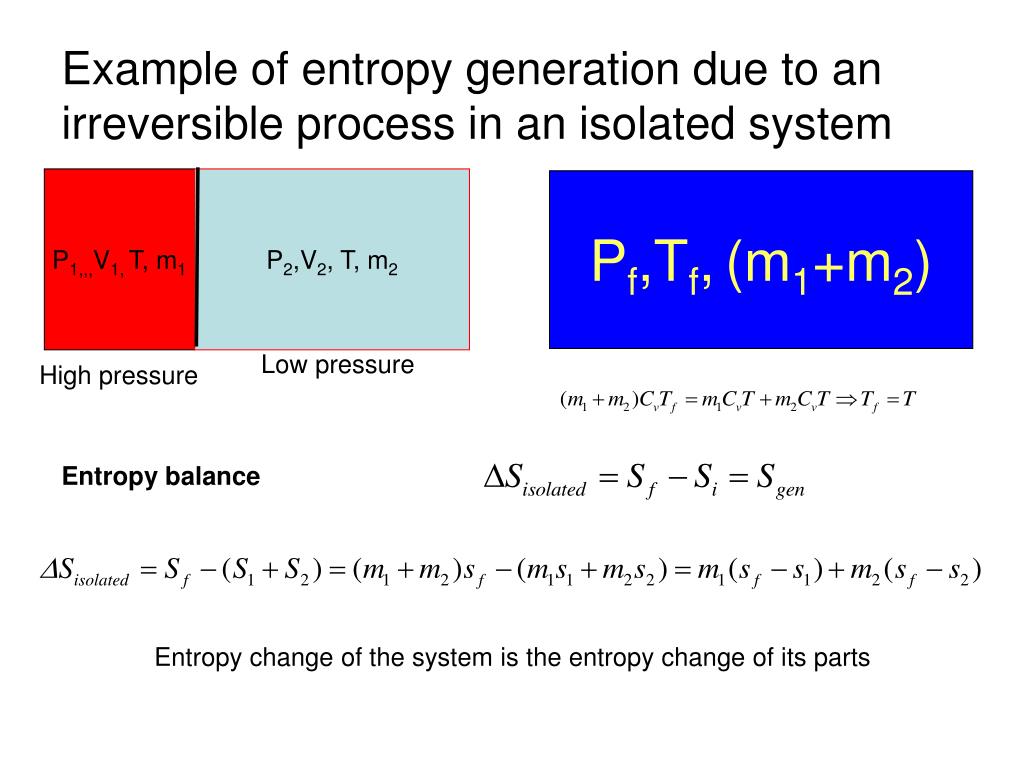
Once you go to systems with order 10^23 coins the predictions how it will evolve get very strong. For example, living systems are clearly able to achieve a local reduction in their entropy as they grow and develop they create structures of greater internal energy (i.e., they lower entropy) out of the nutrients they absorb. Cost Function in a Decision Tree How Does Entropy Actually Work Example of Entropy in Machine Learning Calculation of Entropy in Python Use of Entropy in Decision Tree Conclusion Frequently Asked Questions What is Entropy in Machine Learning In machine learning, entropy measures the impurity or randomness present in a dataset. 
In quantum terms, it is the movement from pure to mixed states, a highly correlated/concentrated state like, all the coins the same way up in a stack, to a mixed state, like each coin is randomly heads or tails spread over the floor. We can move order and disorder around, but in total the disorder increases.Īnother way to describe entropy is to say it's spreading out of energy, the most spread out usually being heat. It's like the ordered bit is the cool part of the fridge - it can't cool the room as a whole, because the fins at the back are releasing a more than balancing amount of heat. Two examples of high entropy and low entropy are: 1) High Entropy: A heater in a ventilated room where Heat flows from Hot to Cold. (4) forsomeconstantC.Thisisanexampleshowingthatentropyisastatevariable:itdependsonlyon thecurrentstateofasystem,nothowitgotthere(heatandworkarenotstatevariables).Clausius formulaassumedentropywasastatevariable.WiththeBoltzmannformula,wecancheck. The Gini index has a maximum impurity is 0.5 and maximum purity is 0, whereas Entropy has a maximum impurity of 1 and maximum purity is 0. Entropy helps explain many of the mysteries and experiences of daily life. Another example is the mixing of ammonium chloride and barium hydroxide, which absorbs. The only way to make things orderly again is to add energy. Yes disorder was created to make the ordered cigarette or tube. For example, CART uses Gini ID3 and C4.5 use Entropy. is obviously product-favored, even though the reaction is endothermic. 
Could you get them back, from those outputs? The key is the whole system. Consider these as closed systems, including the chemical energy involved in squeezing, the light radiated by smouldering. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
